Our new study is out !
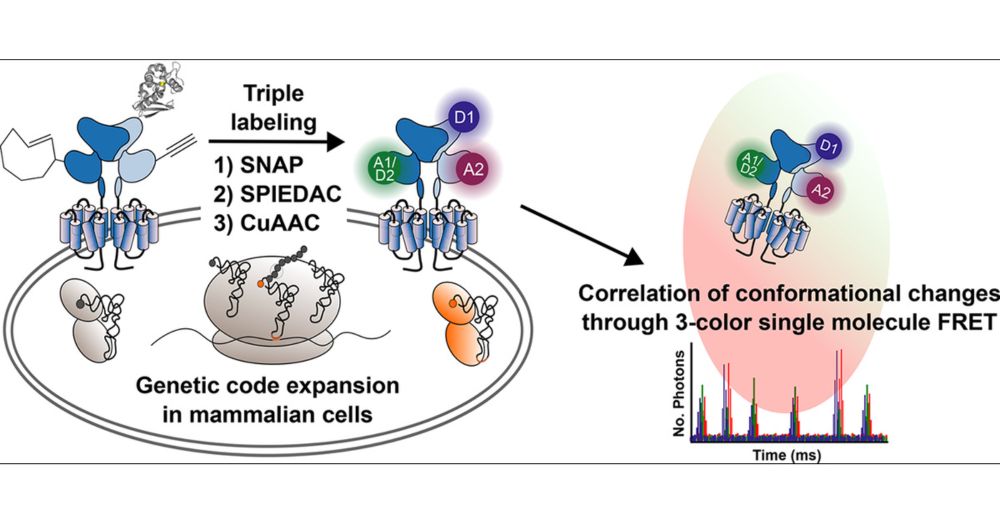
We introduce a triple labeling strategy (2 non canonical Amino Acids + 2 click chemistries and 1 SNAP-tag) to triple label a G-protein coupled receptor.
Open access in JACS
pubs.acs.org/doi/10.1021/...
@rbquast.bsky.social
Biochemistry PI @CBS Montpellier working at the interfaces of biophysics, chemical and structural biology to unravel how structural dynamics control membrane protein function with particular focus on GPCRs.

Our new study is out !
We introduce a triple labeling strategy (2 non canonical Amino Acids + 2 click chemistries and 1 SNAP-tag) to triple label a G-protein coupled receptor.
Open access in JACS
pubs.acs.org/doi/10.1021/...

Postdoc opportunity to elucidate the structural dynamics of GPCRs in our lab euraxess.ec.europa.eu/jobs/309285
22.01.2025 08:30 — 👍 3 🔁 2 💬 0 📌 1
Check our new preprint ! Triple orthogonal labeling of a GPCR with 2 unnatural aminoacids + 1 SNAP-Tag for 3 color #smFRET. We resolve a new intermediate in the activation of metabotropic glutamate receptor 2.
#GPCR #clickchemistry #singlemolecules
www.biorxiv.org/content/10.1...
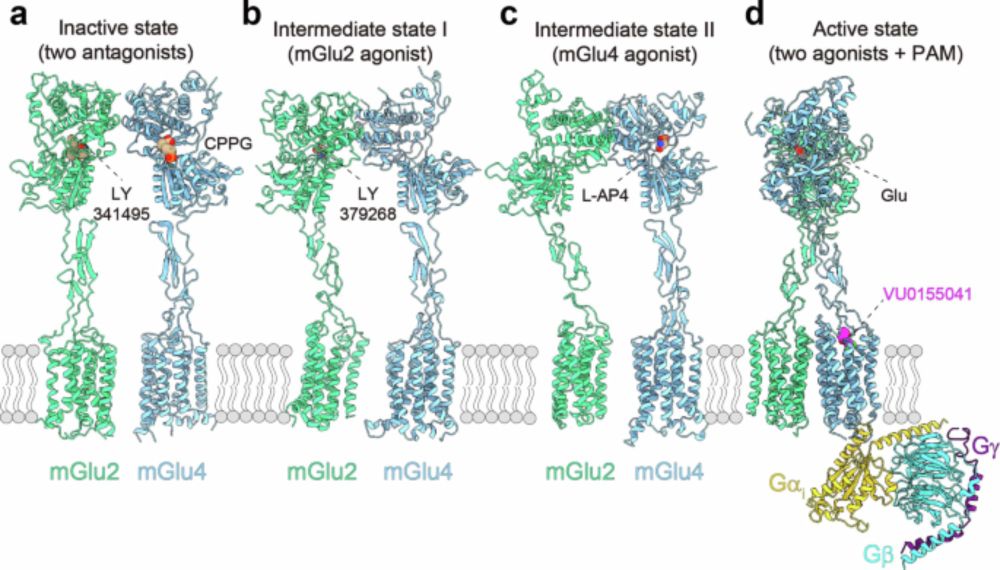
Our new study is out ! We combined #cryoEM and #smFRET to reveal the structural basis of the activation of an heterodimeric #GPCR, the metabotropic glutamate receptor mGluR2-4.
Fantastic collaboration with the teams of Jean-Philippe Pin, Yan Zhang & Jianfeng Liu.
www.nature.com/articles/s41...