Sickness engrams modulate anticipatory immune responses https://www.biorxiv.org/content/10.1101/2025.11.03.686140v1
05.11.2025 16:15 — 👍 7 🔁 5 💬 0 📌 0Sickness engrams modulate anticipatory immune responses https://www.biorxiv.org/content/10.1101/2025.11.03.686140v1
05.11.2025 16:15 — 👍 7 🔁 5 💬 0 📌 0@andreamunozz.bsky.social and I are now based in the Icahn school of Medicine at Mount Sinai in the brilliant labs of @michelenamorado.bsky.social and @mcalpinelab.bsky.social respectively, so keep an eye out for the cool work to come from there also!
24.04.2025 14:42 — 👍 2 🔁 1 💬 0 📌 0This work was supported with funding from @researchireland.bsky.social, @erc.europa.eu and @nihr.bsky.social, and was carried out across multiple institutions including @tbsi-tcd.bsky.social, @tcddublin.bsky.social, @harvard.edu, @princetonmolbio.bsky.social, and @columbiauniversity.bsky.social
24.04.2025 14:41 — 👍 1 🔁 0 💬 1 📌 0A massive thank you to my postdoc mentor @tjryan.bsky.social, amazing lab mates (bluesky-less) Esteban Urrieta, Paul Conway, James O’Leary, @clrar.bsky.social, and brilliant collaborators @lydialynch.bsky.social and @drchristineannd.bsky.social without who this project could not have been done.
24.04.2025 14:36 — 👍 3 🔁 1 💬 1 📌 0
In summary: mice can form memories of cold environments and upregulate metabolism in a non-cold environment as preparation. 🔥
What’s next?
→ Some of the big questions now are whether we can harness these cold memories to treat metabolic dysfunction and disease? 🍔🍟

But wait there’s more! We can turn these cells on, but can we also turn them off?
By using engram specific DREADDs, if we inhibit the DG engram cells, we see no increases in whole-body metabolism.

By labelling neurons in the DG at 4°C, we can later activate them with light at the flick of a switch.🎚️🔋⚡️
Through activating the DG engram cells we find:
📈Whole-body metabolism
📈BAT thermogenesis genes
📈Engrams in the LHA, suggesting connectivity with the DG.
How do we know these cells are ACTUALLY responsible for increasing metabolism in response to memory of a cold environment?
By using lights and lasers of course!💡🔦
Our tagging techniques allow us to express light-sensitive channels on neurons, which we can activate with light.

We can visualise the cells that are important for the cold memory in the DG and LHA.
We show that only the mice that reexperience the previous 4oC environment show increased cell overlap (🟢+🔴=🟡) -> meaning the cells active during the cold, are also active during the memory.
But how do we know that these cells are in fact memory (engram) cells? Using activity dependent labelling we can show:
🟢Cells active at 4oC
🔴Cells active during memory at 21oC
🟡The memory engram – cells which were active during the cold (🟢) and reactive at memory recall (🔴)

Therefore, the memory of a cold environment alone, can increase whole-body metabolism when not in the cold. 🐭💭❄️
What pathways mediate the cold memory brain-body connection? We found increased coordinated activity between the HPC and HY, particularly the DG and LHA.

We found that mice who have previously experienced a cold (4oC) environment will:
📈Increase their whole-body metabolism🔥, and
📈Upregulate heat producing genes in brown fat to stay warm. 🔥
In response to the previously 4oC environment – but in the ABSENCE of the cold (21oC).
We set out to answer 2 questions:
1) Do mice store information in the brain about where they have previously felt a cold environment? ❄️🥶
2) Can accessing the stored information (memory) cause mice to produce more heat? 💭🌡️
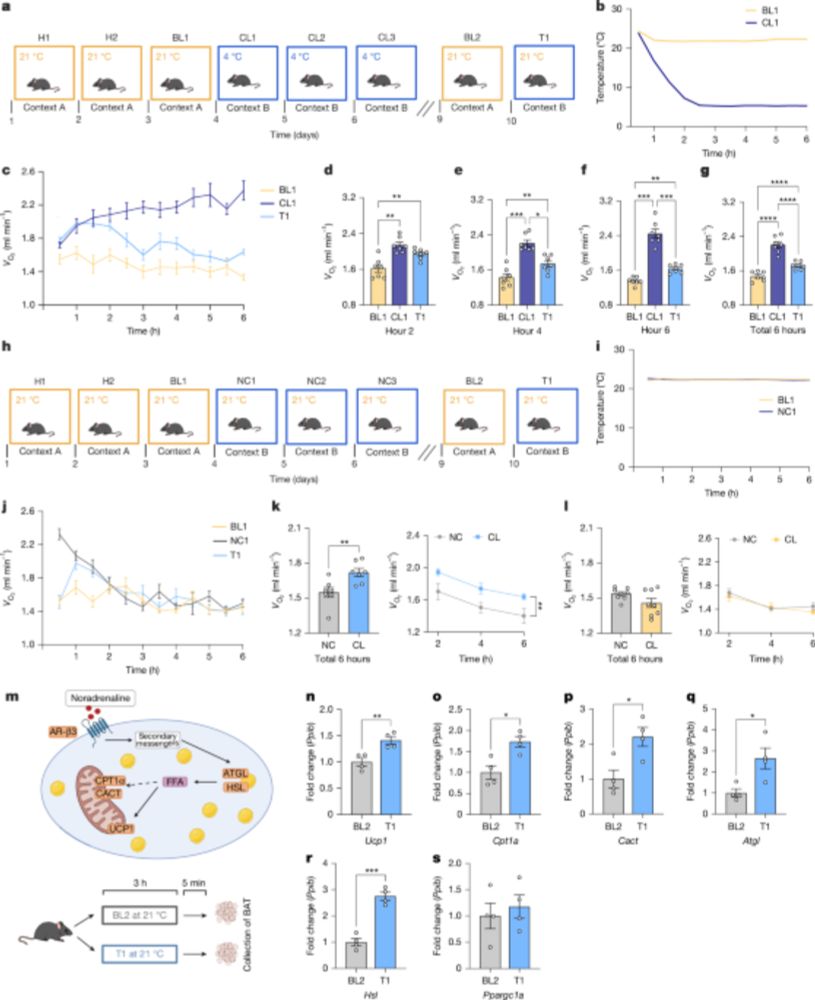
Very excited to share part of my postdoctoral work from the @tjryan.bsky.social lab, out now in @nature.com. Here, my brilliant co-author @andreamunozz.bsky.social and I explored the link between memory engrams and whole-body metabolism.
A thread: 🧵
www.nature.com/articles/s41...