Several Sarpong Group members are featured in a story on College of Chemistry community members with ties to Africa!
chemistry.berkeley.edu/news/chemist...
Several Sarpong Group members are featured in a story on College of Chemistry community members with ties to Africa!
chemistry.berkeley.edu/news/chemist...

Congratulations to Charis, Tenta and Matt, and our collaborator on structural elucidation, Dr. Settineri, on the publication of our total synthesis of hispidospermidin in JACSAu!
pubs.acs.org/doi/10.1021/...

Congratulations to Hiroki on his beautiful syntheses of bis cyclotryptamine alkaloids, now out in JACS! We are grateful to Dr. David Small for his collaboration on the computational aspects!
doi.org/10.1021/jacs...

Congratulations to team "Skeletal Annulation" (Phil, Atang, Antonia, Sam, Nicolo, and Takashi) and our collaborators Pfizer
led by @bagphos.bsky.social on the publication of our work in JACS!
doi.org/10.1021/jacs...
And here it is – Annulative Editing – aka The Monster Mash. What a super collab #PfizerChemistry & @sarponggroup.bsky.social
In 1 pot, activated pyrimidines with bis-nucleophiles like amino-pyrazoles & other small hets to deliver 6/5 hets and other useful products
#MyFirstJACS
#ChemSky #ChemChat

Excited to welcome first years Richard, David P., Nick, and David B. to the group!
06.10.2025 22:49 — 👍 2 🔁 0 💬 0 📌 0
Congratulations to Taku and Pierre on the publication of their dahurelmusin synthesis! Thank you to Dr. Blane Zavesky, our collaborator from Corteva for going above and beyond!
doi.org/10.1021/acs....

Check out this highlight on our carbamorphone work! www.acsh.org/news/2025/08...
14.08.2025 20:10 — 👍 5 🔁 2 💬 0 📌 0
Congratulations to Dr. Richmond Sarpong (@sarponggroup.bsky.social) on being awarded the 2025 Inhoffen Medal.
This award is considered to be "the most prestigious German award in the field of natural products chemistry."
chemistry.berkeley.edu/news/richmon...

This beautiful work from the @sarponggroup.bsky.social also got a nice write-up in @cenmag.bsky.social
01.08.2025 21:10 — 👍 7 🔁 2 💬 0 📌 0
Behold the double-edged sword of natural product medicinal chemistry! On the one hand, doing the SAR is such a pain that industry has largely given up. On the other, look what we haven’t tapped into: a one-atom change to morphine, never before made in the 73 years since the first total synthesis!
01.08.2025 21:07 — 👍 15 🔁 4 💬 1 📌 0
Check out this Article in @cenmag.bsky.social. News on skeletal editing that discusses some of our group's work!
cen.acs.org/synthesis/Sk...

Congratulations to Jack and Safaa on their beautiful study toward the aphidicolin family of diterpenoids that just appeared in JOC!
pubs.acs.org/doi/full/10....

Check out this article in @dailycal.bsky.social highlighting our recently published carbamorphine work!
www.dailycal.org/news/campus/...
Check out this article highlighting our carbamorphine work!
chemistry.berkeley.edu/news/carbamo...

Congratulations to Team Carbamorphine, led by Sota, along with our collaborators from the Majumdar and McLaughlin groups on the publication of our work in PNAS!
www.pnas.org/doi/10.1073/...

A brief editorial with @sarponggroup.bsky.social and @alisonwendlandt.bsky.social where we try to answer an important question: What is Editing?
pubs.acs.org/doi/full/10....

Meet Dr. Sojung Kim, Recent PhD Graduate of UC Berkeley. Headshot of Sojung with C-CAS and UC Berkeley logos

Image of Sojung in front of scientific poster. Caption: "Organic chemists synthesize molecules by connecting atoms or smaller molecule pieces together through chemical reactions. Sojung’s research focuses on using visible light (e.g., colored LED lights) as an energy source to drive new kinds of chemical reactions that ultimately enable us to build a wider variety of molecules more efficiently."

Image of Sojung skiing in Lake Tahoe. Caption: "Sojung grew up in PA and graduated from Penn State as part of the Schreyer Honors College and Millennium Scholars Program. Outside of lab, Sojung loves enjoying the CA sunshine and temperate Bay weather while exploring new restaurants in SF, beach-hopping down the Pacific Coast Highway, wine tasting in Sonoma and Napa, skiing in Lake Tahoe, and tending to her lab balcony garden."
Meet Sojung, a recent graduate of the
@sarponggroup.bsky.social!
Have a questions for Sojung? Let us know!
#NSF #Chemistry #C-CAS #Meetascientist #Chemsky

Congratulations to Sojung, Charis, and Logan on the publication of their Accounts of Chemical Research paper on our group's skeletal editing work, which, at its core, is inspired by total synthesis!
pubs.acs.org/doi/epdf/10....

Check out this @chemistryworld.com highlight of Ryan and Moto's work!
www.chemistryworld.com/news/skeleta...
Chemists at @ucberkeleyofficial.bsky.social led by @sarponggroup.bsky.social figured out how to shuffle peripheral pieces of a molecule by twisting its interior structure, like rearranging a Rubik's cube. Their work, in @science.org, helps speed drug design. chemistry.berkeley.edu/news/twist-c...
09.05.2025 18:23 — 👍 1 🔁 1 💬 0 📌 0
Check out this article highlighting our acyl transposition work!
chemistry.berkeley.edu/news/twist-c...
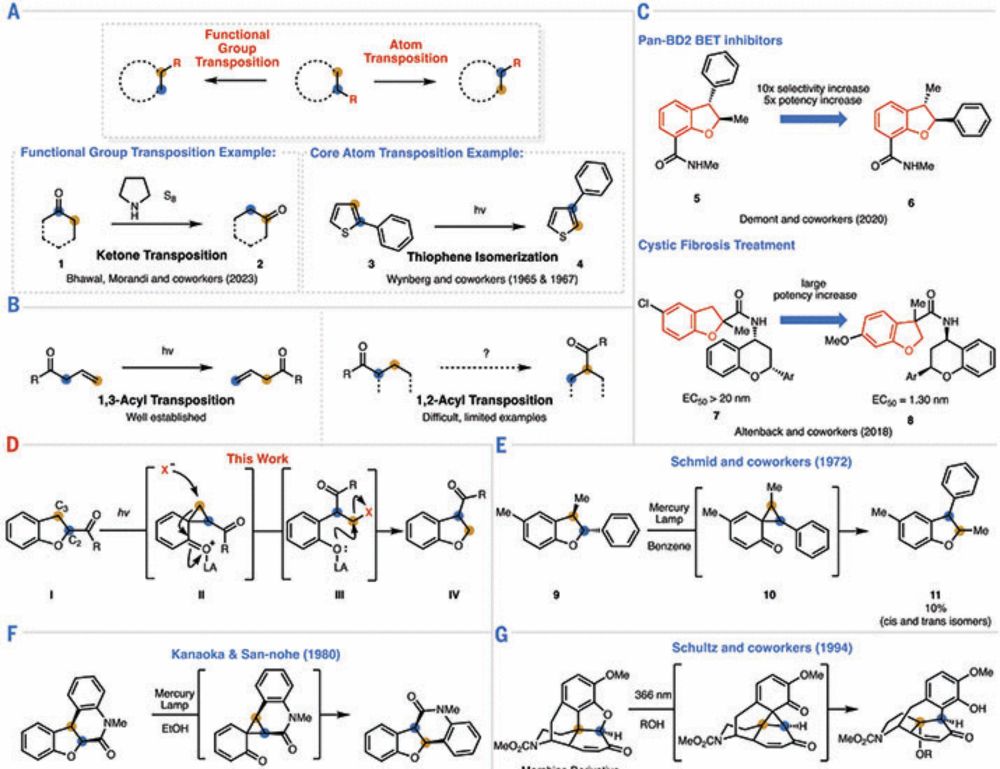
Congratulations to Ryan and Moto on the publication of their 1,2-acyl transposition in 2,3-dihdyrobenzofurans in @science.org!
www.science.org/doi/10.1126/...

In @science.org this week, Ryan Steele and Motohiro Fujiu in @sarponggroup.bsky.social move an acyl group by transposing adjacent carbons rather than moving the oxygen 🔀
chemsky 🧪
www.science.org/doi/10.1126/...

Check out these articles highlighting Richmond's election to the National Academy of Sciences!
news.berkeley.edu/2025/05/02/n...
chemistry.berkeley.edu/news/richmon...

Richmond has been elected into the US National Academy of Sciences!
www.nasonline.org/news/2025-na...

Congratulations to @gwynethpudner.bsky.social, Selena and Eric on the publication of their approach to benzannulated seven-membered rings in @synthesis1969.bsky.social!
www.thieme-connect.com/products/ejo...
Congrats to Nina for being awarded an NSF graduate research fellowship! 🎉 We are so proud of you!
10.04.2025 14:02 — 👍 6 🔁 0 💬 0 📌 0
Meet Suh, a graduate student in the @sarponggroup.bsky.social
Check out our instagram @nsf_ccas to learn more about Suh.
#NSF #Chemistry #C-CAS #Meetascientist

Congrats to Logan & our collaborators Lucas (Sigman Group) & Reilly & @charlesthechemist.bsky.social (Merck) on the publication of our work on a cheminformatic analysis of core-atom transformations in pharmaceutically relevant heteroaromatics. Check it out in J. Med. Chem. now!
tinyurl.com/42re37pd