That would work, thank you!
06.12.2025 15:17 — 👍 1 🔁 0 💬 1 📌 0That would work, thank you!
06.12.2025 15:17 — 👍 1 🔁 0 💬 1 📌 0Is there a preferred term we should use in study materials?
05.12.2025 13:46 — 👍 0 🔁 0 💬 1 📌 0Thank you for inviting me, it was great to hear about your important work! Great effort to develop methods for using real world data in drug development for #ALS #MND, including synthetic controls with the potential to reduce placebo & trial duration!
22.05.2025 19:14 — 👍 1 🔁 0 💬 1 📌 0
@rpavaneijk.bsky.social & colleagues conducted a multi-year prospective cohort study of 97 patients with #ALS. Their findings: an #accelerometer -based measure is predictive of disease progression and related to patient-reported functioning: www.thelancet.com/journals/ebi...
05.03.2025 18:15 — 👍 4 🔁 1 💬 0 📌 0
Our study on the safety and tolerability of Trimetazidine in #ALS @braincomms.bsky.social
Trimetazidine reduced oxidative stress markers and energy expenditure, warranting a follow up study
@rpavaneijk.bsky.social @fredsteyn.bsky.social @ammaralchalabi.bsky.social
academic.oup.com/braincomms/a...

Dropout & attrition in #ALS #MND trials is a major challenge. We have created an #open-access calculator to estimate attrition rates for a given study duration, which can used to guide trial design and sample size calculations (tricals.shinyapps.io/Attrition/); the accompanying paper: rdcu.be/d3n36
13.12.2024 10:15 — 👍 8 🔁 1 💬 0 📌 0Yes as prognostic factor it works great, could be used as eligibility criteria to leave out slow progressors, but correlation NFL - progression is not 1 to 1 and comparative data is sparse yet how well this works (and how much better it is than simpler methods based on FRS/clinics).
10.12.2024 16:46 — 👍 0 🔁 0 💬 1 📌 0Regarding NFL, most programs include it as secondary objective now. I have some reservations still around NFL as probably small effects like Riluzole will not show a large change and would be difficult to assess in non-randomised studies.
10.12.2024 16:25 — 👍 1 🔁 0 💬 1 📌 0You are right, the riluzole example is an RCT for patients who can't participate in the main RCT, which I believe is similar for EAP (only those not eligible for trial?). Think you need some control to make the data insightful.
10.12.2024 16:25 — 👍 2 🔁 0 💬 1 📌 0Thanks! I would be interested in the starter pack as well!
10.12.2024 15:48 — 👍 1 🔁 0 💬 1 📌 0
Maybe redesign how we use EAPs. I like the approach riluzole used - they did a controlled EAP study, which provided useful data (pubmed.ncbi.nlm.nih.gov/12021952/): "The primary objective was to enable access to treatment to patients excluded from the pivotal trial which was run in parallel."
10.12.2024 15:41 — 👍 4 🔁 0 💬 2 📌 0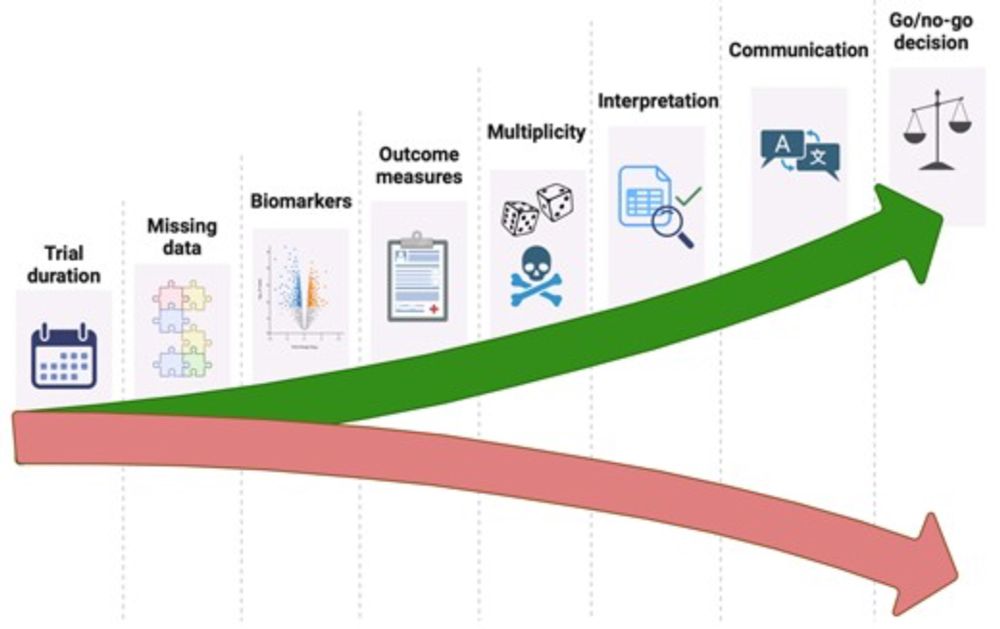
New paper to reflect on the main challenges in phase 2 #ALS #MND trials. Key issues relate too short study durations, reliance on the wrong outcome measures, absence of biomarkers, statistical issues, over-interpretation of 'clinical trends' and miscommunication. academic.oup.com/brain/articl...
10.12.2024 15:14 — 👍 18 🔁 7 💬 6 📌 4Good points; EAP could evaluate safety and tolerability, but it is difficult to determine drug benefit in EAP. OLE (or ATE - active treatment extension - as I was told over the weekend) holds value to get some efficacy data, especially if there is a delayed effect, but doesn't replace a placebo.
10.12.2024 15:00 — 👍 2 🔁 0 💬 1 📌 0