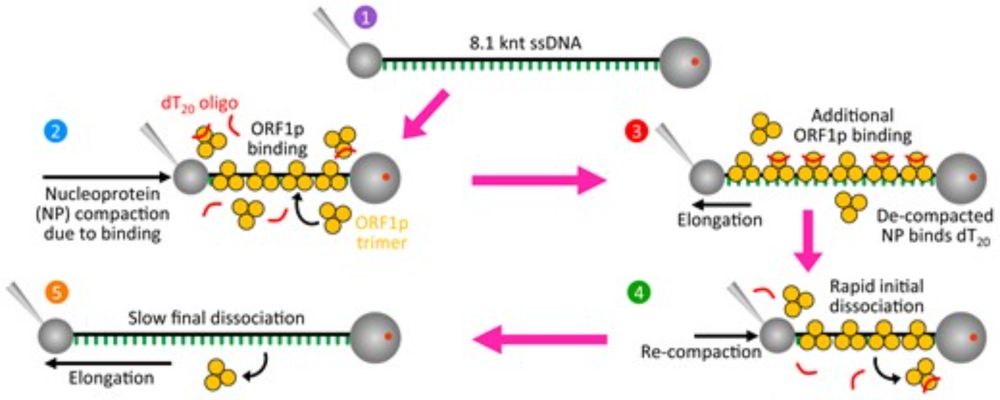
How does one protein do two jobs? Our new study shows how phosphorylation changes SARS-CoV-2 nucleocapsid from irreversibly compacting RNA (for packaging) to fast on/off binding (for replication). Part of a collaboration with the Musier-Forsyth lab at Ohio State. Link: doi.org/10.3390/v18010105
13.01.2026 17:52 — 👍 3 🔁 1 💬 0 📌 0Mark Williams single molecule biophysics lab
@williamslabneu.bsky.social
Optical tweezers | AFM | correlated fluorescence and force | nucleic acid-protein interactions | SSBs | chromatin stability | nucleosome chaperones | retrovirus, coronavirus, retrotransposon replication https://williamslab.sites.northeastern.edu