The light at the back is shining on a bank of LEDs connected in parallel to provide a very small current but enough to microelectrolyse 0.5M CuCl₂ solution. Electrodes are carbon rods.

Chlorine reacts with KI solution. It turns blue litmus red and bleaches. It,causes starch/iodate paper to turn blue with the formation of iodine

After 10 minutes, litmus paper is almost bleached but slightly red, and the starch/iodate paper is almost white. And now 0.5M KBr has turned slightly yellow with the formation of bromine water.

After 10 minutes, the cathode shows a little copper. A 9V battery would bleach litmus in 2 minutes and show more copper, as the current would be larger.
Electrolysis of 0.5M CuCl₂ solution powered by light! NOT A BATTERY. See, the electrolysis is so slow that the litmus is actually red before bleaching. LED banks are in parallel, better than the previous tweet on this topic.
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
01.02.2026 22:57 —
👍 4
🔁 1
💬 2
📌 0

The Beer's law plot is very good. A spreadsheet is used to find the values.

Usual values are in the 67% region.

There is absorption in the UV and blue area and the yellow-to-red region. This gives chlorophyll its green colour.

Increasing the iodine concentration does not increase the rate of reaction. Increasing the acid and propanone concentrations increases the rate of reaction.
Here is a Beer's Law plot of copper nitrate solution used to find the copper content of brass. The different coloured LEDs can show the absorption spectrum of chlorophyll. The rate of reaction of the iodination is the final slide.
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
01.01.2026 22:58 —
👍 1
🔁 1
💬 0
📌 0

The new design is top left. The Lego cuvette holder is top right. The voltmeter is the most expensive part.

The downside is that the absorbance has to be calculated. However, a lof money has been saved for a set of 10.

There are 6 coloured LEDs and a UV LED in the experiments. Is it any good? See the next message.
Innovative design moves with new technology. The cuvette holder moves from Lego to a 3D-printed one. It uses a coloured LED as the emitter and an IR-LED as the collector. This produces a voltage related to the transmitted light.
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
01.01.2026 22:58 —
👍 3
🔁 1
💬 1
📌 0
See how little solid is used to do this. The main video is on www.youtube.com/watch?v=FWxT....
It only takes a minute, allowing time for drawing mechanisms. Why does it form in the middle? Why does it sparkle? How is it yellow?
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
24.12.2025 19:10 —
👍 4
🔁 1
💬 0
📌 0
Yes it much nicer to use than cabbage. Here is the pH profile. Watch my lockdown project on butterfly pea tea and more. See www.youtube.com/watch?v=tbR5...
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
23.12.2025 14:27 —
👍 7
🔁 0
💬 0
📌 0

This is electrolysis using light. The 8 light cells are arranged in parallel to provide a voltage of about 3 V and a current of 65 mA. Copper chloride solution is electrolysed to give copper and chlorine Bleaching litmus, affecting starch/iodide paper, and potassium iodide solution.

Sustainable electrolysis using light instead of wasting the battery. Copper on the cathode
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
13.12.2025 22:55 —
👍 4
🔁 1
💬 0
📌 0
This has a beauty of its own when projected on a screen in a lecture theatre. A diffusing complex forms when Fe(NO₃)₃ and KCNS come together. Look what happens as an Nd magnet comes close. "In a little you can see a lot."
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
07.12.2025 22:33 —
👍 12
🔁 1
💬 1
📌 0

13ISMC26
Wednesday 8 July to Friday 10 July 2026
Go to sites.google.com/view/13ismc2....
It's about micro- and small-scale practical activities inspired by the school educators in chemistry around the world. Look at the gallery page! Sign up on the front page. Send a proposal
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
06.12.2025 14:10 —
👍 4
🔁 2
💬 1
📌 0
Try these in a 1-1.5 cm diameter droplet of water on a polypropylene surface. Why do this? A short activity leads to a long discussion on ionic lattices, dissolving (solvation), diffusion, precipitation & spectator ions.
#chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
05.12.2025 15:59 —
👍 8
🔁 3
💬 0
📌 0
Watch the ppt appear. Ca(NO₃)₂ goes in late, as it dissolves quicker. It's better to get the ppt close to the centre. This process occurs in less than a minute. It works for any precipitation, but this is a "green" version. #chemsky #chemchat #realtimechem #beyondbenign #iTeachChem #Chemistry
04.12.2025 20:11 —
👍 15
🔁 3
💬 0
📌 0
Here is the video of the glistening crystals of lead iodide. The glistening crystals are formed during formation (as they are so thin) and by the bombardment with surrounding water molecules. #ChatChemistry, #ITeachChem @cleapss.bsky.social
#chemistry #greenchemistry @rsc-eic.bsky.social
24.10.2025 08:03 —
👍 9
🔁 1
💬 1
📌 0

Tiny speck of Pb(NO₃)₂ was introduced on the left (you can still see the remains) in a 1 cm diameter of water; I waited 10 seconds and put a tiny speck of KI from the right.
What a fantastic lead iodide precipitate by diffusion I obtained yesterday. The sparkling changes were beautiful.
24.10.2025 06:59 —
👍 5
🔁 0
💬 0
📌 0
We are comparing the diffusion speeds of halogens along damp starch/iodide paper. This activity is conducted in Petri dishes on the bench. The final section shows iodine result and uses 1 tiny ball of iodine.
#ChatChemistry, #ITeachChem @cleapss.bsky.social #chemistry #greenchemistry
19.10.2025 10:52 —
👍 6
🔁 2
💬 0
📌 0

Thanks. The purple vapour is visible evaporation. Other solids evaporate, such as ice in a freezer and camphor and menthol at room temperature. I hope you have made a note of this.
18.10.2025 21:35 —
👍 2
🔁 0
💬 0
📌 0
Confusion abounds as to whether iodine sublimes or evaporates at 1 atm. In fact, it melts and flows. A little microscale triumph. See tinyurl.com/2nw2hram
#ChatChemistry, #ITeachChem
@cleapss.bsky.social @rsc-educomscot.bsky.social @sfed-rsc.bsky.social @beyondbenign.bsky.social
18.10.2025 19:38 —
👍 4
🔁 3
💬 1
📌 0

This is separating activated carbon from an aqueous suspension.
You can watch this method of filtering on youtu.be/6rSu-blfRZo. This is separating activated carbon from an aqueous suspension. Carbon was used to remove a dye.
02.08.2025 14:34 —
👍 1
🔁 0
💬 0
📌 0

Atmospheric chemistry is difficult for schools, but microscale techniques can help with evidence for ozone and hydroxyl radicals. Here are the videos referred to in the picture.
youtu.be/mlQRDuKm63w
www.youtube.com/watch?v=U_PA...
www.youtube.com/watch?v=9jNh...
#ITeachchem
26.05.2025 19:03 —
👍 1
🔁 0
💬 0
📌 0
And there was I, thinking you sat down and watched these videos with pen in hand eager for all the information
22.05.2025 10:40 —
👍 0
🔁 0
💬 1
📌 0
if you go to 13 seconds in the video, there is a QR code to a supplier. I hope that helps
21.05.2025 21:49 —
👍 1
🔁 0
💬 1
📌 0
Here is a diffusing precipitate of silver iodide on a thermochromic plastic sheet so that the endothermic dissolving and the exothermic precipitation are seen.
#ITeachchem #RealTimeChem #chatchemistry
@sfed-rsc.bsky.social @beyondbenign.bsky.social,
bsky.app/profile/cerg...
21.05.2025 11:37 —
👍 5
🔁 3
💬 1
📌 1
YouTube video by Bob Worley
IronIII thiocyanate complex
Watch for the effect of a magnet on the iron/thiocyanate complex. Paramagnetism at work.
www.youtube.com/watch?v=qipp...
#ITeachchem #RealTimeChem #chatchemistry @sfed-rsc.bsky.social
01.03.2025 20:24 —
👍 11
🔁 7
💬 0
📌 2

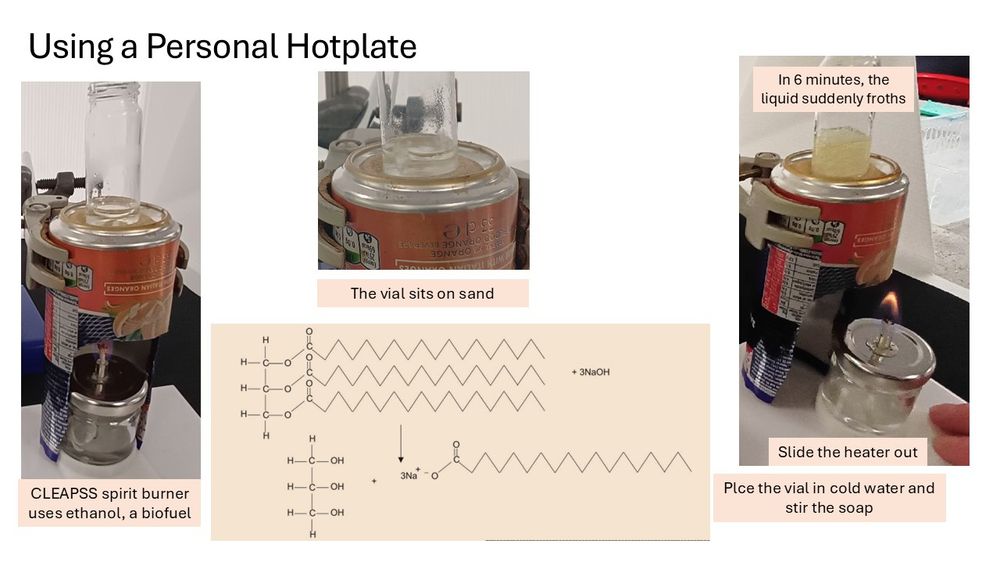
0.25 cm³ of 1M copper sulfate is added to the iron staples and 2 cm³ of 1.5M sulfuric acid It is heated for 10 minutes. Can also use steel paper clips. This is the personal spirit burner (ethanol biofuel) hotplate.

Using filter paper in small-scale chem does not work. Filtering the reaction mixture through cotton wool in a Pasteur pipette. Copper particles and bits of iron are separated from the iron(II) sulfate solution..

Heating the filtered solution for 10 minutes on my personal spirit-burner, bio-fuel, hotplate.
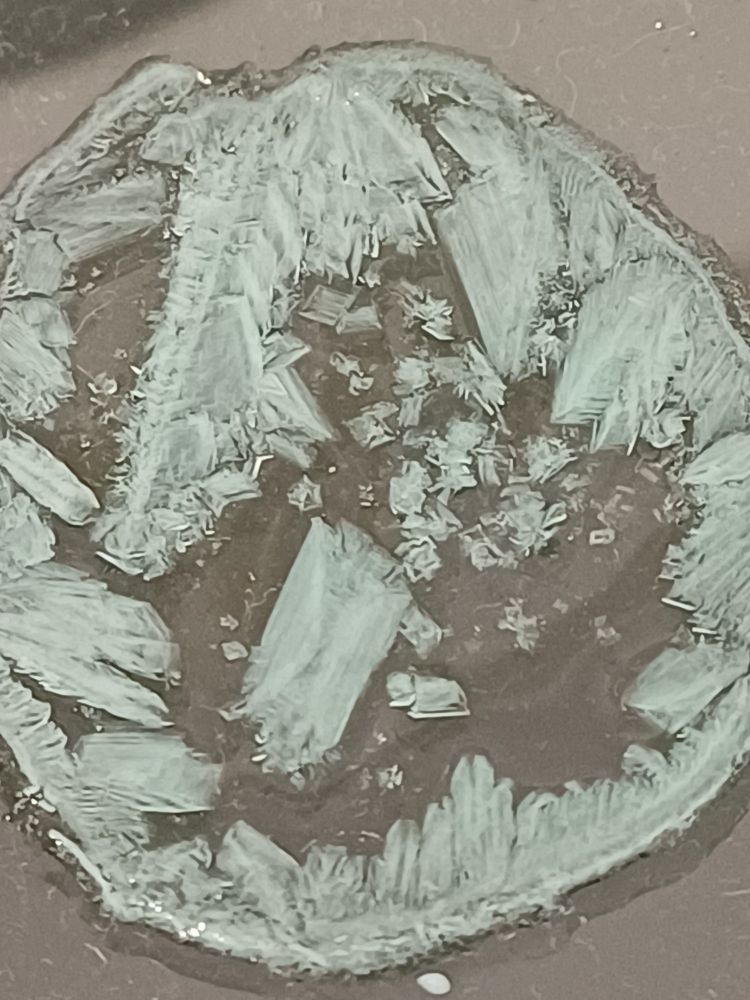
Then leave to cool. The crystals form slowly, which might take longer than a lesson, but the results are very beautiful.
Helplines asked #CLEAPSS for an alternative to CuO/H₂SO₄ neutralisation reaction salt making for students to make salts. Not easy, but using ed/ox metal/acid reactions uses the same skills. This is iron(II) sulfate made from staples.
#ITeachchem #RealTimeChem #chatchemistry
22.02.2025 12:28 —
👍 7
🔁 2
💬 0
📌 0
Making 1-5% soap solutions in 7:3 propan-2-ol/water. Filtered to get a colourless liquid. Laser light reflection indicates it is colloidal. Is the twinkling effect caused by collisions with water molecules?
#ChatChemistry #ITeachChem @cleapss.bsky.social @cerg.bsky.social
06.12.2024 06:27 —
👍 12
🔁 2
💬 0
📌 1

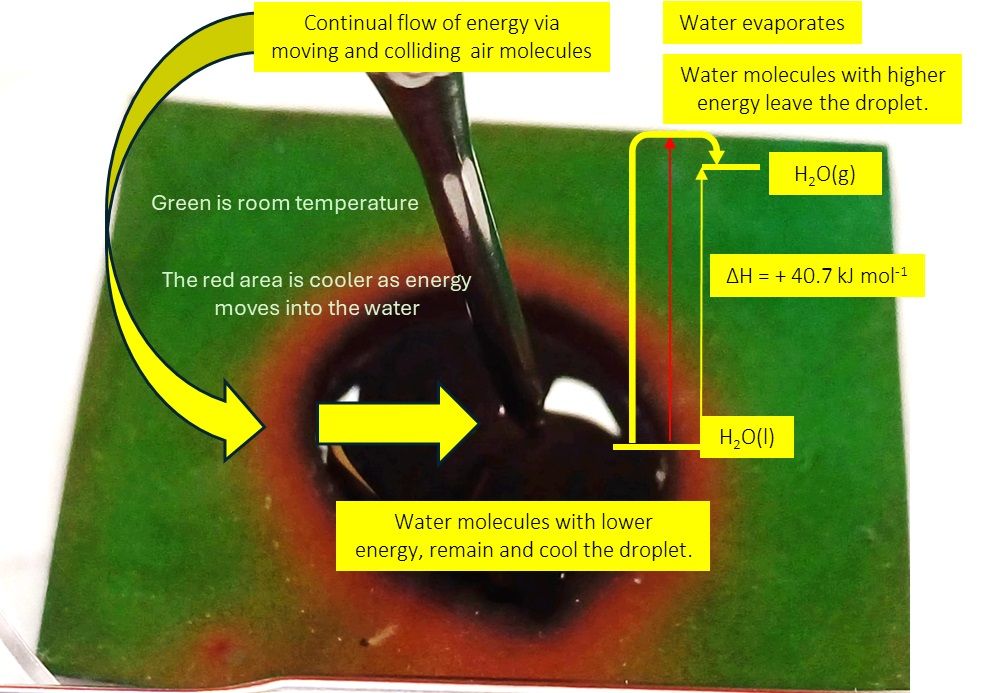
Evaporation with Thermochromic plastic and 2 kitchen thermometers. Visualise the flow of energy in an open endothermic process at room temperature—an evidence-based small-scale approach using easily available modern materials. #ChatChemistry #ITeachChem @sfed-rsc.bsky.social @cerg.bsky.social
26.11.2024 06:13 —
👍 5
🔁 1
💬 1
📌 0
It works better with bottle top crucible. See Cleapss website. Do a comparison of methods if you want.
14.11.2024 12:50 —
👍 1
🔁 0
💬 1
📌 0

Five go to Thailand
This is the link to www.rsc.org/news-events/... for an account of the chemistry education trip to Thailand. @bogwoppitfee.bsky.social #ChatChemistry, #ITeachChem @cleapss.bsky.social @associationscied.bsky.social @roysocchem.bsky.social @beyondben.bsky.social , @unitednationsbk.bsky.social
09.10.2024 17:18 —
👍 6
🔁 2
💬 0
📌 0
It is all about energy. I think chemists are rather scared of the topic. We invent octet rules to explain and ditch it. We are awe of physics teaching.
02.10.2024 12:14 —
👍 1
🔁 0
💬 1
📌 0