Effective Recycling Pathways of Commodity Polymers Enabled by Mechanoradical Capture
Plastics pervade every aspect of modern life, yet effective mechanical recycling remains a major challenge. This is, in part, because of the mechanical forces that are involved in reprocessing, which break polymer chains and generate mechanoradicals, leading to a reduction in molecular weight and diminished material properties. This work introduces a robust strategy to capture and redirect these reactive intermediates, enabling value-preserving recycling pathways for widely used polymers polystyrene (PS) and poly(methyl methacrylate) (PMMA). By employing ball milling to induce chain scission, we demonstrate that mechanoradicals can be trapped by bis(butyl trithiocarbonate), yielding polymers with trithiocarbonate (TTC) end groups. Polymers degraded via ball milling showed significant reduction in molecular weight, ≈90% lower than the pristine polymers. These low molecular weight, TTC-functionalized polymers then served as macroinitiators for light-mediated controlled polymerization or, in the case of PMMA, as mediators for depolymerization under mild conditions. Chain extension of the degraded materials led to restored or increased molecular weight compared to the pristine polymers. Shear oscillatory rheology experiments revealed a recovery of entangled polymer properties, as evidenced by the reappearance of the rubbery plateau. We further showed that this “capture-and-repair” strategy is compatible with multiple cycles of degradation and chain extension, achieving repeated molecular weight recovery over three cycles. Additionally, we found that ball milling alone lowers the thermal depolymerization temperature of PMMA, enabling up to ≈44% depolymerization at 220 °C. Together, these findings highlight mechanoradical capture as a promising strategy to both enhance circularity and improve overall performance of mechanically recycled plastics.
Very excited to share our recent article in JACS where we showed that we could capture mechanoradicals formed during polymer degradation and use them to grow polymers back to high MWs or prime them for depolymerization! pubs.acs.org/doi/10.1021/...
10.11.2025 19:59 —
👍 6
🔁 4
💬 0
📌 0
A busy mentor taking time to reach out to a potential PI can be very impactful. It doesn't happen often. It's especially helpful if you can provide some texture that might not be evident from the applicant's resume and transcript.
24.09.2025 21:28 —
👍 5
🔁 0
💬 1
📌 0
The New York Times piece today about US science is terrible and wrong—in many ways.
I could write a whole article about this, but as one example:
“To close observers, the original crisis began well before any of this…”
No. I’m a close observer of science, and this is incorrect.
22.09.2025 12:20 —
👍 915
🔁 224
💬 24
📌 30

Really proud of Riley's preprint on a new class of chiral Lewis acid photocatalysts. This project benefitted from a terrific collab with @chemguyeli.bsky.social chemrxiv.org/engage/chemr...
22.08.2025 15:12 —
👍 36
🔁 4
💬 2
📌 0
DNA-Scaffolded Ultrahigh-Throughput Reaction Screening
Discovering and optimizing reactions is central to synthetic chemistry. However, chemical reactions are traditionally screened using relatively low-throughput methods, prohibiting exploration of diver...
Excited to share our new preprint, which was years in the making! chemrxiv.org/engage/chemr...
New reactions are typically developed by trial and error. How can we speed up this process? Read on to learn how we used DNA scaffolding to perform >500,000 parallel reactions on attomole scale.
1/n
14.08.2025 17:40 —
👍 40
🔁 19
💬 1
📌 3
I don't perceive this crisis in Chemistry - not as a reviewer, editor, or author. Sure I'd like it if I got a couple fewer requests each month and if my reviewers accepted a couple more invitations apiece - but neither constitutes a hashtag-level issue.
30.07.2025 23:12 —
👍 10
🔁 1
💬 0
📌 0


Last night, the 2025 Roger Adams Award recipient - Prof. Eric Jacobsen - delivered a fantastic lecture at the National Organic Symposium. Prof. John Wood provided a brilliant introduction.
The award is sponsored by @OrgReactions and @OrgSynth
Congratulations!
25.06.2025 13:37 —
👍 6
🔁 2
💬 0
📌 0

PhD position in machine learning for photocatalysis
We are recruiting a PhD student in machine learning for photocatalysis! In this project, we will collaborate with the group of Frank Glorius @gloriusgroup.bsky.social to develop predictive tools for energy-transfer-catalyzed photocycloadditions.
Reposts appreciated!
jobs.ethz.ch/job/view/JOP...
13.05.2025 08:15 —
👍 30
🔁 19
💬 1
📌 2
Spring 25' saw the kick-off of our first #MerckChemistry Symposium at UW-Madison. We were blessed with beautiful weather and an amazing day of science. Special thanks to our hosts @tehshik.bsky.social & Shannon Stahl, Scott and Emma as Merck organizers, and the entire UW Chem and ChemEng community!
12.05.2025 20:11 —
👍 2
🔁 1
💬 1
📌 0
Like shh man.
29.04.2025 23:33 —
👍 7
🔁 0
💬 0
📌 0
US Applicant Week
U.S. citizen interested in grad studies at the University of British Columbia in Vancouver (Canada)?
Grad programs are re-opening applications of US programs for one week. With expedited decisions.
U.S. Applicant Week:
www.grad.ubc.ca/us-applicant...
#AcademicChatter #Canada #GradSchool
07.04.2025 19:49 —
👍 426
🔁 332
💬 9
📌 23
#teamroslyn
19.03.2025 16:55 —
👍 1
🔁 0
💬 0
📌 0
Dani!!! That is so awesome!!!
08.03.2025 21:10 —
👍 1
🔁 0
💬 0
📌 0
"No no, nobody calls me Dr Freeze, Dr Freeze is my dad. Call me Todd."
28.02.2025 16:39 —
👍 3
🔁 0
💬 1
📌 0
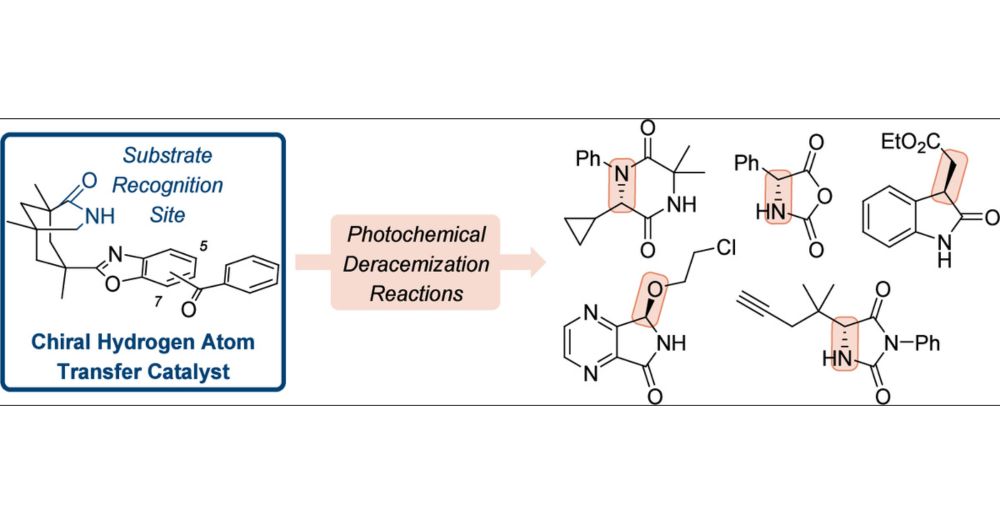
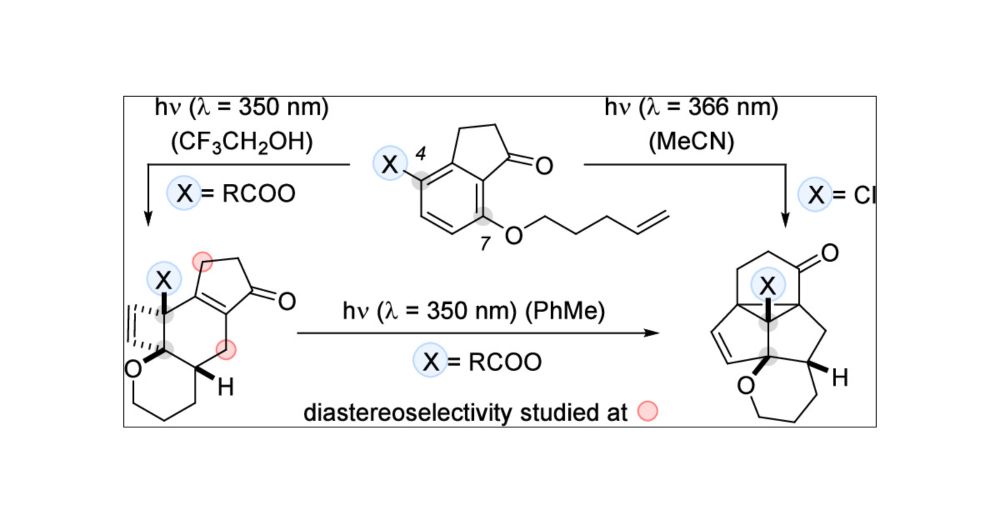
Stereochemical Editing at sp3-Hybridized Carbon Centers by Reversible, Photochemically Triggered Hydrogen Atom Transfer
ConspectusMillions of chiral compounds contain a stereogenic sp3-hybridized carbon center with a hydrogen atom as one of the four different substituents. The stereogenic center can be edited in an increasing number of cases by selective hydrogen atom transfer (HAT) to and from a photocatalyst. This Account describes the development of photochemical deracemization reactions using chiral oxazole-annulated benzophenones with a bonding motif that allows them to recognize chiral lactam substrates by two-point hydrogen bonding. The backbone of the catalysts consists of a chiral azabicyclo[3.3.1]nonan-2-one with a U-shaped geometry, which enables substrate recognition to occur parallel to the benzoxazole part of the aromatic ketones. The photocatalysts facilitate a catalytic photochemical deracemization of several compound classes including hydantoins, N-carboxyanhydrides, oxindoles, 2,5-diketopiperazines, and 4,7-diaza-1-isoindolinones. In addition, if more than one stereogenic center is present, the editing delivers a distinct diastereoisomer upon the appropriate selection of the respective photocatalyst enantiomer. The chiral photocatalysts operate via the benzophenone triplet that selectively abstracts a properly positioned hydrogen atom in exclusively one of the two substrate enantiomers. The photochemical step creates a planar carbon-centered radical and erases the absolute configuration at this position. While returning HAT to the same position would likely recreate the stereogenic center with the same absolute configuration, spectroscopic and quantum chemical studies suggest that the hydrogen atom is delivered from the photocatalyst to a heteroatom that is in conjugation to the radical center. Two scenarios can be distinguished for the hydrogen atom shuttling process. For hydantoins, N-carboxyanhydrides, and 4,7-diaza-1-isoindolinones, the back HAT occurs to a carbonyl oxygen atom or an imine-type nitrogen atom which is not involved in binding to the catalyst. For oxindoles and 2,5-diketopiperazines, a single lactam carbonyl group in the substrate is available to accept the hydrogen atom. It is currently assumed that back HAT occurs to this group, although the carbonyl oxygen atom is involved in hydrogen bonding to the catalyst. In comparison to the former reaction pathway, the latter process appears to be less efficient and more prone to side reactions. For both cases, an achiral enol or enamine is formed, which delivers upon dissociation from the catalyst statistically either one of the two stereoisomers of the substrate. Since only one substrate enantiomer (or diastereoisomer) is processed, a high enantioselectivity (or diastereoselectivity) results. Even though the editing is a contra-thermodynamic process, the described decoupling of a photochemical and a thermal step allows the usage of a single catalyst in loadings that vary between 2.5 and 10 mol % depending on the specific mode of action.
Interested in stereochemical editing by photochemically triggered hydrogen atom transfer? Check out our latest review by Maxi 💡🧪
pubs.acs.org/doi/10.1021/...
25.02.2025 16:40 —
👍 9
🔁 1
💬 0
📌 0
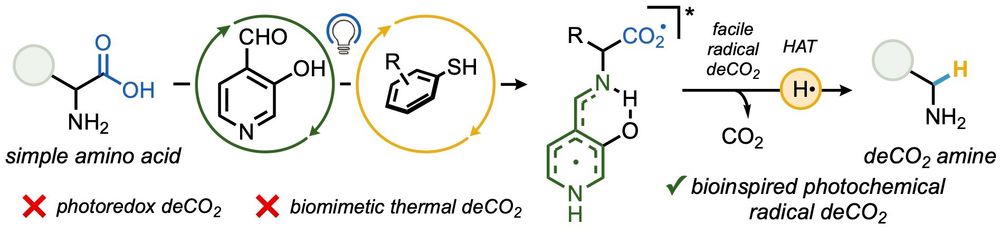
🚨 NEWS 🚨 Super excited to share that our first paper has been published in @angewandtechemie.bsky.social, describing our development of Vitamin B6-inspired amino acid photodecarboxylation! doi.org/10.1002/anie...
19.02.2025 15:49 —
👍 17
🔁 3
💬 1
📌 1

Newton International Fellowships | Royal Society
This fellowship is for non-UK scientists who are at an early stage of their research career and wish to conduct research in the UK.
📢 Royal Society Newton International Fellowship applications open now, to host international postdocs in the UK! Are you interested in a fellowship in photochemistry, heterocycles and/or strained molecules? Email me with your CV & details of your research interests 👍 royalsociety.org/grants/newto...
15.02.2025 18:24 —
👍 1
🔁 2
💬 0
📌 0
Second!
29.01.2025 01:08 —
👍 2
🔁 0
💬 1
📌 0
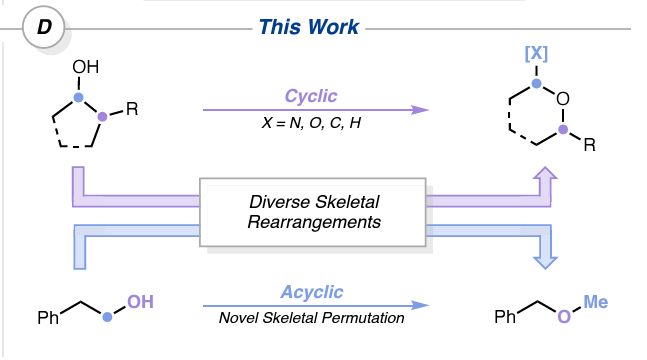
Really excited to see this amazing work out in ChemRxiv from our #ChemCollab between the @uwyoongroup.bsky.social @tehshik.bsky.social and #PfizerChemistry
Photochem-mediated oxygen migration into sp3 bonds delivering small ring ethers as well as an OH - methylene transposition
#ChemSky 1/
23.01.2025 00:37 —
👍 34
🔁 8
💬 4
📌 0

oxygen and nitrogen insertion reactions into saturated carbocycles
Friends, I'm pleased to draw your attention to two new preprints today describing complementary heteroatom insertion reactions into saturated carbocycles, one from our group and another from the Knowles laboratory.
chemrxiv.org/engage/chemr...
chemrxiv.org/engage/chemr...
22.01.2025 16:40 —
👍 62
🔁 12
💬 4
📌 3
What will really annoy chemists is the Texas carbon….
21.01.2025 14:05 —
👍 25
🔁 2
💬 4
📌 0
Academic Young Investigator's Symposium (YAS) - ACS Division of Organic Chemistry
The Academic Young Investigator's Symposium is for Asst. Prof.'s who are entering their 5th or 6th years and who have not yet been considered for tenure.
Hey Organic Asst Profs coming up for tenure (and your senior colleagues) — it is nomination season for the ACS DOC Young Academic Investigators Symposium. Deadline is Feb. 6, 2025. The website has last year’s date 😳 but the other info is right. #ChemSky www.organicdivision.org/meetingsuppo...
14.01.2025 19:17 —
👍 10
🔁 11
💬 0
📌 0