This was a wonderful collaboration with Sophia Fochler, Eva Gluenz, @zephyris-science.bsky.social, @alanbrownhms.bsky.social, and more analyzing a mountain of data. We are also grateful for the support through a dual NSF and Swiss NSF grant.
10.11.2025 17:45 —
👍 2
🔁 0
💬 0
📌 0
Want to know more without the sports analogy? Check out the full story on bioRxiv www.biorxiv.org/content/10.1...
10.11.2025 17:45 —
👍 2
🔁 0
💬 1
📌 0

We also knocked out individual doublet microtubule subunits, revealing which structural elements are important for movement. Together, our work provides a new framework for understanding how diverse molecular “rowers” coordinate ciliary motility.
10.11.2025 17:45 —
👍 1
🔁 0
💬 1
📌 0
Next, we systematically deleted each dynein gene and analyzed how these knockouts altered flagellar movement. The results were surprising: each dynein distinctly impacted motility, but not necessarily in the ways predicted from earlier studies.
10.11.2025 17:45 —
👍 1
🔁 0
💬 1
📌 0
Using Leishmania as a model, we determined the cryo-EM structure of the doublet microtubule to pinpoint the position of each dynein. This gave us a detailed map of where every “rower” sits on the ciliary “boat.”
10.11.2025 17:45 —
👍 4
🔁 2
💬 1
📌 1

In an eight-person rowing boat, each rower contributes to movement but also has a unique role: balancing, powering, or setting the rhythm and pace. In our latest collaborative work we asked – do the eight dynein “rowers” in #cilia and #flagella operate in the same way?
10.11.2025 17:45 —
👍 11
🔁 5
💬 1
📌 1

A conserved mechanism for the retrieval of polyubiquitinated proteins from cilia
The temporospatial distribution of proteins within cilia is regulated by intraflagellar transport (IFT), wherein molecular trains shuttle between the cell body and cilium. Defects in this process impair various signal-transduction pathways and cause ciliopathies. Although K63-linked ubiquitination appears to trigger protein export from cilia, the mechanisms coupling polyubiquitinated proteins to IFT remain unclear. Using a multidisciplinary approach, we demonstrate that a complex of CFAP36, a conserved ciliary protein of previously unknown function, and ARL3, a GTPase involved in ciliary import, binds polyubiquitinated proteins and links them to retrograde IFT trains. CFAP36 uses a coincidence detection mechanism to simultaneously bind two IFT subunits accessible only in retrograde trains. Depleting CFAP36 accumulates K63-linked ubiquitin in cilia and disrupts Hedgehog signaling, a pathway reliant on the retrieval of ubiquitinated receptors. These findings advance our understanding of ubiquitin-mediated protein transport and ciliary homeostasis, and demonstrate how structural changes in IFT trains achieve cargo selectivity. ### Competing Interest Statement The authors have declared no competing interest. Sara Elizabeth O'Brien Trust Postdoctoral Fellowship awarded through the Charles A. King Trust Postdoctoral Research Fellowship Program, , 8460873-01 Richard and Susan Smith Family Foundation, https://ror.org/05j95n956, National Institute of General Medical Sciences (NIGMS), , R01GM141109, R01GM143183
How do cells keep their cilia “clean” and functional? Our new study uncovers a conserved mechanism for retrieving polyubiquitinated proteins from #cilia – a process essential for cellular signaling and health. #cellbiology #ciliopathy #ubiquitin #IFT 🧵👇 1/n
29.04.2025 15:04 —
👍 54
🔁 26
💬 2
📌 2
Finally this work wouldn’t be possible without the team
in the Brown lab @alanbrownhms.bsky.social
13.03.2025 21:31 —
👍 1
🔁 0
💬 0
📌 0
This was a fantastic collaboration with @ruizhangmt.bsky.social, Richard Wheeler, and Eva Gluenz. Check out the paper for more!
13.03.2025 21:22 —
👍 2
🔁 0
💬 1
📌 0
It is also important to highlight that this work was made possible by funding through multiple NIH and NSF grants. These funding mechanisms are vital for biomedical research.
13.03.2025 21:22 —
👍 1
🔁 0
💬 1
📌 0
Our strategy of combining structural analysis with comprehensive genetic knockout, promises to continue uncover the mechanisms that control flagella-based motility.
13.03.2025 21:22 —
👍 1
🔁 0
💬 1
📌 0

We also observe trypanosomatid-specific axoneme specializations. One example is the B-tubule ponticulus structure, which was first observed nearly 60 years ago! We find that the lumen-spanning structure is made up of three components, whose periodicity is established by a filamentous MIP.
13.03.2025 21:22 —
👍 2
🔁 0
💬 1
📌 0

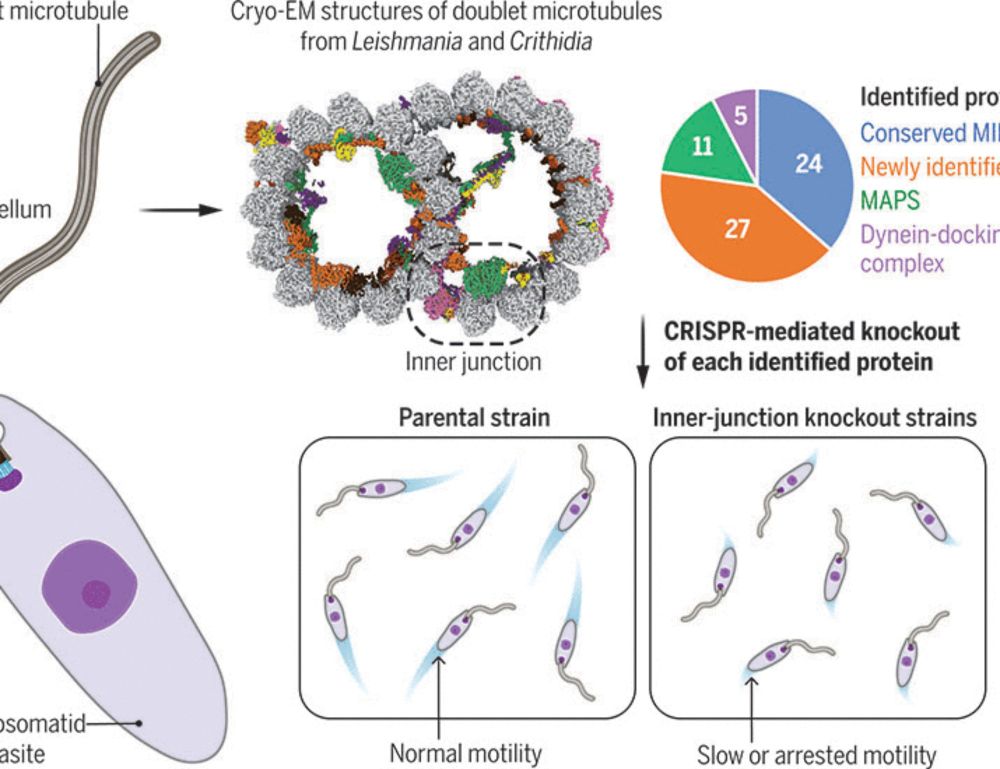
Using CRISPR, we knocked out each of our identified proteins and tested the mutant swimming speed. Our analysis found that the doublet is surprisingly resilient to individual MIP knockout. However, we show that the evolutionarily conserved inner junction is uniquely sensitive to knockout.
13.03.2025 21:22 —
👍 1
🔁 0
💬 1
📌 0

Our structure revealed a highly specialized doublet containing 51 microtubule inner proteins (MIPs). Once resolved, we used Leishmania as a model to test the contribution of each MIP to motility.
13.03.2025 21:22 —
👍 2
🔁 0
💬 1
📌 0