Phosphorylation-driven signaling plays a central role in how cells communicate. In this study, together with @saezlab.bsky.social , we revisit the EGF signaling pathway and ask: How well does our traditional understanding hold up in the era of modern phosphoproteomics? www.nature.com/articles/s41...
03.03.2026 14:57 —
👍 12
🔁 6
💬 1
📌 0
A comprehensive understanding of protein-driven signaling is not just conceptually satisfying. It is essential for better drugs, understanding resistance, and building “virtual cells”; a vision that will remain out-of-reach if we neglect protein-driven signaling.
02.03.2026 12:35 —
👍 1
🔁 0
💬 0
📌 0
My take? Advancing our knowledge of signaling pathways will require scalable, unbiased, and reproducible perturbational phosphoproteomics, something that is slowly arriving but has not yet reached the scale of Perturb-seq transcriptomic atlases.
02.03.2026 12:35 —
👍 1
🔁 0
💬 1
📌 0
Progress in our knowledge of signaling may require challenging core assumptions about how it works. Is phosphorylation-driven signaling a fine-tuned, precise engine? Or more of an explosion that searches for the activation or inhibition of transcriptional programs?
02.03.2026 12:35 —
👍 0
🔁 0
💬 1
📌 0
These findings suggest kinase-driven signaling is far less mapped than we assume. Maybe not all phosphosites are equally informative, and many reported kinase–substrate interactions may be less robust than we think. Current maps likely mix missing edges with questionable ones.
02.03.2026 12:35 —
👍 0
🔁 0
💬 1
📌 0
The result: ~90% of kinase–kinase interactions in the resulting networks are not part of any current ground truth set. Are they biologically real and relevant, or artifacts of incomplete knowledge and noise? We still don’t know, but the gap is hard to ignore.
02.03.2026 12:35 —
👍 0
🔁 0
💬 1
📌 0
In this paper, we focused on EGF signaling and asked: how many kinase–kinase interactions, consistent with phosphoproteomics data and prior knowledge, belong to a bespoke “ground truth”? We benchmarked across datasets, resources, and “ground truth” definitions.
02.03.2026 12:35 —
👍 0
🔁 0
💬 1
📌 0
One of the main stories of my postdoc is finally out! It builds on an intuition from my early days in omics data analysis: our signaling pathways are simply too small, underrepresenting cellular complexity after stimulation. Something obvious, yet surprisingly hard to quantify.
02.03.2026 12:35 —
👍 8
🔁 3
💬 1
📌 0


10 years and still standing strong @EMBL 🍾💪
Huge thanks to all members, alumni, supporters & colleagues who made it possible.
Here’s to the next decade! 🙏
24.02.2026 10:38 —
👍 47
🔁 5
💬 0
📌 2
Happy to share new work led by @miraburtscher.bsky.social, together with colleagues from @savitski-lab.bsky.social, @saezlab.bsky.social (shout-out to @martingarridorc.bsky.social), @zimmermannlab.bsky.social and others.
Original post + paper below! 👇
12.02.2026 10:43 —
👍 9
🔁 4
💬 0
📌 0
When I first learned about omics at university, I confidently stopped caring about single proteins. Fast-forward 4 years of a system’s biology PhD, I now have the most amazing favorite single protein which I can’t stop thinking about. Here is the story of how that happened 👀
11.02.2026 14:06 —
👍 15
🔁 6
💬 0
📌 0

Rewiring of oncogenic signaling in #DrugResistance is a moving target. In our new study, we used biophysical phosphoproteomics to investigate #BRAF mutant cancer, linking phosphorylation changes to protein function and #MolecularMechanisms through #Multi-Omics integration.
tinyurl.com/funsignaling
11.02.2026 13:57 —
👍 29
🔁 12
💬 1
📌 3
Gracias Victoria!
27.01.2026 19:06 —
👍 0
🔁 0
💬 1
📌 0
Muy contento de estar de vuelta en casa para las #JABI2026. ¡Gracias por la invitación!
----
Happy to be back home for #JABI2026. Thanks for the invitation!
27.01.2026 10:29 —
👍 3
🔁 0
💬 0
📌 0

Want to learn about the theoretical aspects of proteins/peptides analysis by mass spectrometry?
Then join the #EMBLProteomics course where you will get hands-on experience in sample preparation and perform tandem mass tag labelling.
Apply by 3 March:https://s.embl.org/sdp26-01-bl 🔬💻
03.12.2025 09:32 —
👍 4
🔁 3
💬 0
📌 0
New preprint 🚨 We systematically measured 17 million phospho-specific dose-response curves (133 kinase inhibitors × 5 cell lines) to decrypt the kinases that shape the human phosphoproteome. We show that drug perturbation potency (not effect size) links kinases to substrates while controlling FDR.
20.11.2025 11:59 —
👍 22
🔁 7
💬 0
📌 0
Finally out! 🤩 Check out our HT- PELSA for high throughput screening!
05.11.2025 16:28 —
👍 16
🔁 6
💬 1
📌 1
Happy to see our HT-PELSA paper now published in @natsmb.nature.com 🎊 Big thanks for the constructive review process! 📖Read the manuscript here (www.nature.com/articles/s41...) & check the thread for additional information ⬇️
05.11.2025 10:15 —
👍 37
🔁 14
💬 0
📌 1

🚨 New preprint
We present an extended version of ScAPE, the method that won one of the prizes 🏆 in the @neuripsconf.bsky.social 2023 Single-Cell Perturbation Prediction challenge.
📄 preprint: doi.org/10.1101/2025...
🧬 code: github.com/scapeML/scape
19.09.2025 07:45 —
👍 30
🔁 7
💬 1
📌 1
Our 2023 SCP model is now out as a manuscript! Curious about a lightweight baseline for your context generalization task? Check it out 👇
19.09.2025 11:02 —
👍 2
🔁 0
💬 0
📌 0
ScAPE: A lightweight multitask learning baseline method to predict transcriptomic responses to perturbations https://www.biorxiv.org/content/10.1101/2025.09.08.674873v1
09.09.2025 19:47 —
👍 2
🔁 2
💬 0
📌 0

So grateful and honored to receive the MCP Lectureship Award at the #ASBMBProteomics meeting at the Broad Institute! Huge thanks to the wonderful organizers and to everyone—past and present—who’s been part of our lab.
20.08.2025 10:09 —
👍 42
🔁 7
💬 2
📌 0

Beeswarm plot of the prediction error across different methods of double perturbations showing that all methods (scGPT, scFoundation, UCE, scBERT, Geneformer, GEARS, and CPA) perform worse than the additive baseline.

Line plot of the true positive rate against the false discovery proportion showing that none of the methods is better at finding non additive interactions than simply predicting no change.
Our paper benchmarking foundation models for perturbation effect prediction is finally published 🎉🥳🎉
www.nature.com/articles/s41...
We show that none of the available* models outperform simple linear baselines. Since the original preprint, we added more methods, metrics, and prettier figures!
🧵
04.08.2025 13:52 —
👍 126
🔁 57
💬 2
📌 6
This project has been in the making for quite some time. CORNETO not only integrates key concepts and methodologies in biological network inference, but also introduces a novel framework for multi-condition analysis. Congrats to the team, and especially to @pablormier.bsky.social for leading this.
24.07.2025 07:51 —
👍 7
🔁 3
💬 0
📌 0

🎉 The revised version of CORNETO, our unified Python framework for knowledge-driven network inference from omics data, is published in peer reviewed form
🔗 Paper: www.nature.com/articles/s42...
📖 News & Views: www.nature.com/articles/s42...
💻 Code: corneto.org
🧵 Thread 👇
22.07.2025 15:23 —
👍 61
🔁 29
💬 1
📌 1
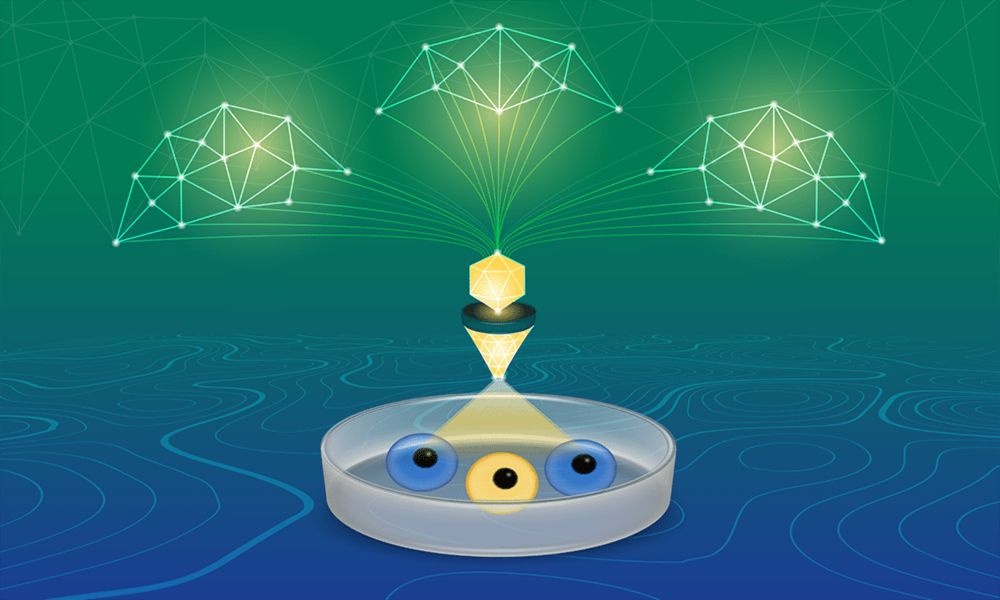
CORNETO: machine learning to decode complex omics data
New tool combines biological knowledge with machine learning to help researchers extract meaningful insights from complex omics data.
How can we find out what’s really going on inside cells when we’re generating so much complex data?
CORNETO is an open-source tool that uses machine learning to turn tangled omics datasets into clear maps of how genes, proteins, and signalling pathways interact.
www.ebi.ac.uk/about/news/r... 🧪
22.07.2025 09:20 —
👍 38
🔁 10
💬 1
📌 0

The final version of our multi-omics study on kidney fibrosis is out now (tinyurl.com/kidneyfibMSB). Together w/ Pepperkok + Savitski labs @embl.org, we present a time-resolved #multiomics + computational network modeling approach in combination w/ phenotypic assays to study #kidneyfibrosis
27.06.2025 13:29 —
👍 22
🔁 11
💬 1
📌 1













