
I remember seeing the americium crystals from like 20 ft away. Great job Nick! #fblockrocks www.nature.com/articles/s41...
20.08.2025 16:10 — 👍 0 🔁 0 💬 0 📌 0
I remember seeing the americium crystals from like 20 ft away. Great job Nick! #fblockrocks www.nature.com/articles/s41...
20.08.2025 16:10 — 👍 0 🔁 0 💬 0 📌 0
@hbwineinger.bsky.social wrote this review for those interested in low valent f elements #fblockrocks www.sciencedirect.com/science/arti...
25.07.2025 18:39 — 👍 12 🔁 5 💬 0 📌 0


Neptunium (Np3+) has a beautiful alexandrite effect. Left is one lab, center is natural sunlight, and right is a lab under fluorescent lighting. 5f-5f transitions are magical. This is the same solution. Some great pics taken by @jp_brannon #fblockrocks #actinides
13.06.2025 03:01 — 👍 2 🔁 0 💬 0 📌 0
249Bk (t1/2 ~330 days) is a beta blasting bully, but sometimes things work out #fblockrocks #bkallday @teasactinides.bsky.social pubs.acs.org/doi/10.1021/...
10.06.2025 14:50 — 👍 9 🔁 3 💬 0 📌 0
Plutonium chemistry is wack and this compound is another reason to provide evidence towards that #fblockrocks @teasactinides.bsky.social
pubs.rsc.org/en/content/a...

This is a heavy hitting americium structure great job brian! @teasactinides.bsky.social pubs.acs.org/doi/10.1021/...
12.03.2025 22:42 — 👍 7 🔁 1 💬 0 📌 0
I am super proud of this paper. It was a massive, multi-year undertaking by three different groups. If you've ever wanted to understand the high-pressure behavior of curium, here it is. @gagliardilaura.bsky.social #fblockrocks #nuclear #chemsky #bluesci #highpressure
pubs.acs.org/doi/full/10....

I'm excited to see this one published! Samarium(II) dibenzo-24-crown-8 complexes from solutions of MeCN, THF, and DME.
pubs.acs.org/doi/10.1021/...

I love pictures of clean californium. This small quantity has enough gamma radiation to give you your annual background dose in ~30 minutes on contact pictured here is the oxalate #fblockrocks
08.01.2025 00:43 — 👍 6 🔁 2 💬 0 📌 0
What an awesome way to start the year!
pubs.rsc.org/en/content/a...

This article was years in the making. Did you think water could coordinate to Sm(II) without being reduced? Well it can. Congratulations Todd! #Chemsky #Bluesci #samarium #rareearths #fblockrocks
pubs.acs.org/doi/10.1021/...
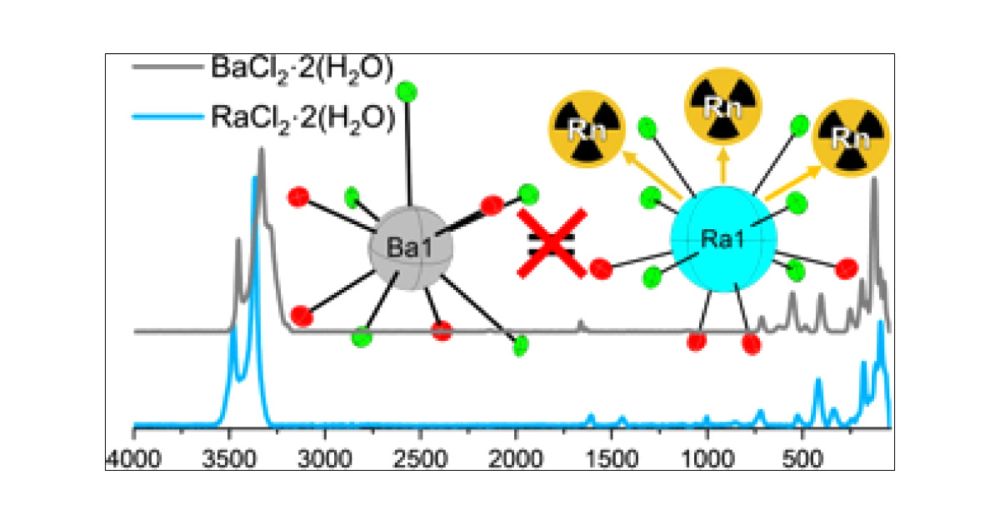
Super happy with this article. Ra-226 makes Cf-249 look cold. There's more radium chemistry to come after the holidays. #Chemsky #bluesci #radium #nuclear #fblockrocks
pubs.acs.org/doi/full/10....

Hi bluesky, meet blueplutonium #fblockrocks
03.12.2024 20:49 — 👍 3 🔁 0 💬 0 📌 0