More than a message: Death by a 1,000 “Chats”
Abstract. Secure electronic messaging within the electronic health record (EHR), such as Epic Chat, has transformed communication in hospitals by enabling
Check out our article and guidance on Epic chat messages in CID. Anyone doing frontline clinical work understands the substantial downsides of constant, on-demand, text communication in the EHR. So great to write this with ID fellows. doi.org/10.1093/cid/...
14.02.2026 15:46 —
👍 28
🔁 11
💬 1
📌 1
Oh nooo! NSW is about to switch to Epic. Any tips on how it works with ID consults welcome!
15.02.2026 08:44 —
👍 1
🔁 0
💬 1
📌 0

My top 10 ID papers for 2025. Full citations and descriptions available at clarityinitiative.org/hot-takes
Also see/hear our Communicable podcast episode on this at communicable.transistor.fm/episodes/com...
What were your top 10? Any thoughts about what I missed welcome!
#IDSky #Top10papers
05.02.2026 23:28 —
👍 31
🔁 9
💬 1
📌 1

📣 New #Communicable drops Mon 26 Jan! In our last episode, we brought you some favorite #clinmicro papers of 2025. We continue this thread in our next episode with top ID papers of 2025, picked by our editors, Josh Davis & Emily McDonald and guest, Steven Tong. ⭐ Tune in Monday🎙️
#IDSky
22.01.2026 10:45 —
👍 8
🔁 5
💬 0
📌 1
It's not syndromic surveillance. It's swabbing every week regardless of symptoms. We also ran metagenomics on those with symptoms but no virus detected - found some interesting things (future paper). We hope to get this funded as an ongoing sentinel surveillance system.
02.01.2026 00:55 —
👍 1
🔁 0
💬 0
📌 0

How Do We Win? The GAME CHANGER Trial
Erin K. McCreary, PharmD (1), Ahmad Mourad, MD MHS (2, 3)
The 3rd and 4th commentaries on recent ID trials are up on the CLARITY website (we aim to publish in depth reviews of important ID stuies).
This time looking at the GAMECHANGER trial:
clarityinitiative.org/commentaries...
@erinmccreary.bsky.social with Ahmad Mourad
@gurujosh.bsky.social
#IDSky
01.01.2026 23:50 —
👍 13
🔁 5
💬 0
📌 0
Congratulations to the RSI trial team #CCRdownunder
10.12.2025 00:34 —
👍 4
🔁 2
💬 2
📌 0
Safety, tolerability, and pharmacokinetics of a 2 g subcutaneous dose of ceftriaxone as an alternative to intravenous delivery
ABSTRACTSubcutaneous delivery of antibiotics is a practical alternative to intravenous administration. Ceftriaxone is commonly used for a variety of infections with limited data on the safety and pharmacokinetics of a 2 g subcutaneous dose. This was a prospective, self-controlled cross-over study in 20 stable inpatients receiving ceftriaxone for their infection. Following an intravenous dose, participants received a single dose of 2 g subcutaneous ceftriaxone, in 50 mL normal saline via gravity feed. Capillary dried blood spots were collected at baseline, 1, 2, 4, 8, and 24 hours following the subcutaneous and intravenous doses. Pain scores and infusion site reactions (edema/erythema) were assessed. Ceftriaxone concentrations were measured using a validated liquid chromatography-tandem mass spectrometry assay. A population pharmacokinetic model was developed using nonlinear mixed-effects modeling. The highest median (interquartile range) pain score within the first 2 hours following infusion of a subcutaneous dose of 2 g ceftriaxone was 2.5 (1–4). All participants were pain-free 4 hours after the infusion. The estimated bioavailability was 95.7% (95% bootstrap interval 90.3–99.5). Compared with intravenous, subcutaneous administration resulted in lower peak and comparable trough concentrations. The probability of target attainment for free drug concentrations was similar to intravenous administration for most common infections in hospitalized patients. Subcutaneous administration of 2 g ceftriaxone is well tolerated and has a comparable pharmacokinetic profile relative to intravenous dosing in non-critically ill patients with severe infections.IMPORTANCEThis prospective, self-controlled cross-over design study demonstrates that subcutaneous administration of 2 g ceftriaxone appears safe and well tolerated with a comparable pharmacokinetic profile relative to intravenous dosing in non-critically ill patients with severe infections.CLINICAL TRIALSThis study was registered at ACTRN12624000692538.
Study of 20 pts showed 2g SC ceftriaxone had 95.7% bioavailability, lower peak but similar trough vs IV; pain score max 2.5, pain-free by 4h; safe & well tolerated.💉🦠
#idsky
01.12.2025 10:30 —
👍 1
🔁 1
💬 0
📌 0
Parliament just passed into law the independent Australian Centre for Disease Control 🎉
The interim CDC will now move from under the Department of Health, Ageing, and Disability to an independent entity as of 1 Jan 2026!
#auspol #idepi #episky #medsky #idsky
06.11.2025 07:21 —
👍 9
🔁 2
💬 0
📌 0
#IDSky #PedsID
27.10.2025 20:27 —
👍 8
🔁 2
💬 0
📌 0
Great to speak with @thelancetinfdis.bsky.social together with @gurujosh.bsky.social about adaptive platform trials in infectious diseases!
23.10.2025 11:38 —
👍 13
🔁 8
💬 1
📌 0
Sorry dud link. See reply
23.10.2025 04:39 —
👍 1
🔁 0
💬 0
📌 0
How long do labs need to incubate #PJI specimens for? Clinicians and some guidelines want long (10-14 days) BUT with modern media and processes, this is probably not needed. See our new paper here sciencedirect.com/science/articl…
@#IDSky
21.10.2025 20:38 —
👍 9
🔁 0
💬 2
📌 0
Big congrats Amy and Richie!
20.09.2025 09:58 —
👍 1
🔁 0
💬 0
📌 0
Let me know if your fellows have any questions for the authors!
04.07.2025 02:03 —
👍 3
🔁 0
💬 0
📌 0
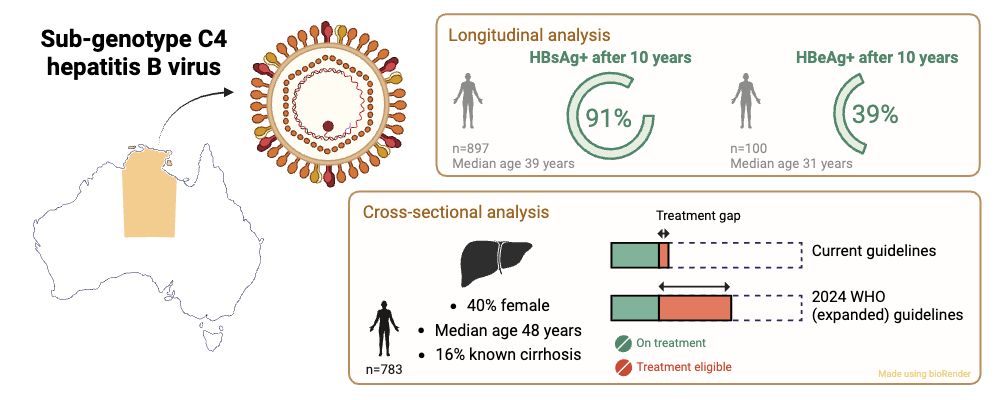
Thrilled to share our new research led by Jane Davies @menziesresearch.bsky.social on First Nations people in the NT living with chronic hepatitis B. An important study that could inform better care:
doi.org/10.1186/s128...
#idsky #MedSky #HepSky #LiverSky
02.07.2025 01:31 —
👍 6
🔁 4
💬 2
📌 0

Communicable E28: Late-breaker trials at ESCMID Global: Should they change your practice? - part 2
Editors of CMI Comms, Josh Davis, Erin McCreary and Emily McDonald...
🎙️ Just published a new #Communicable episode: Communicable E28: Late-breaker trials at ESCMID Global: Should they change ...
Hosted by Josh Davis @gurujosh.bsky.social
Erin McCreary @erinmccreary.bsky.social
Emily McDonald @dremilymcd.bsky.social
Listen here:
#IDSky #Clinmicro #MedSky
01.06.2025 22:01 —
👍 14
🔁 8
💬 0
📌 1

Communicable E27: Late-breaker trials at ESCMID Global: Should they change your practice? - part 1
This episode of Communicable takes on a special format where editors of...
🎙️ Just published a new #Communicable episode: Communicable E27: Late-breaker trials...
Hosted by Marc Bonten @marcbonten.bsky.social
Josh Davis @gurujosh.bsky.social
Erin McCreary @erinmccreary.bsky.social
Emily McDonald @dremilymcd.bsky.social
Listen here:
#IDSky #Clinmicro #MedSky
18.05.2025 22:01 —
👍 12
🔁 8
💬 0
📌 1
⏰ Tune in Monday for the latest #Communicable and hear the complete discussion w/ our editors on some of the late breaker trials at #ESCMIDGlobal '25, part 1! 🎙️
@gurujosh.bsky.social @dremilymcd.bsky.social @erinmccreary.bsky.social @marcbonten.bsky.social @escmid.bsky.social
#IDSky #clinmicro
16.05.2025 06:30 —
👍 12
🔁 4
💬 1
📌 2

Has been great interacting with >850 orthopods and ID doctors from >50 countries at the www.icmortho.com meeting. Lots of interest in the #ROADMAPtrial!
10.05.2025 14:54 —
👍 5
🔁 0
💬 0
📌 0

SNAP Out of It: Rethinking Antistaphylococcal Penicillins for S. Aureus Bacteremia – The SNAP Trial PSSA/MSSA Results
Podcast Episode · Breakpoints · 05/04/2025 · 1h 11m
such a joy to discuss PSSA/MSSA domains of SNAP. incredible work led by @gurujosh.bsky.social @steventong.bsky.social conversation w/ the fabulous @angelahuttner.bsky.social
joint effort @escmid.bsky.social Communicable & @sidpharm.bsky.social Breakpoints podcast
podcasts.apple.com/us/podcast/s...
05.05.2025 16:08 —
👍 20
🔁 8
💬 1
📌 0
The Frank-Starling law of Selective Digestive Decontamination
More than hundred years ago a series of experiments demonstrated that increased strechting
of heart muscles (through higher venous return) increased stroke volume (cardiac output).
But not endlessly, ...
Debated for 40 years: Selective Decontamination of the Digestive tract in ICU patients (antibiotics to prevent ICU-acquired infections). My editorial to Josh Davis @gurujosh.bsky.social and Allen Cheng @peripatetical.bsky.social pro-con in @cmicomms.bsky.social.
www.cmi-comms.com/article/S295...
09.05.2025 06:01 —
👍 12
🔁 4
💬 0
📌 1
Yes. I think it’s mostly because X has mostly (but not completely) died for ID stuff and Bluesky is still not working as well as Twitter did at its peak
15.04.2025 17:56 —
👍 7
🔁 0
💬 1
📌 0

Taking a break from talking about #SNAPtrial to work on a new collaboration
#IDSky #ESCMIDglobal
@steventong.bsky.social
14.04.2025 15:19 —
👍 19
🔁 0
💬 2
📌 0