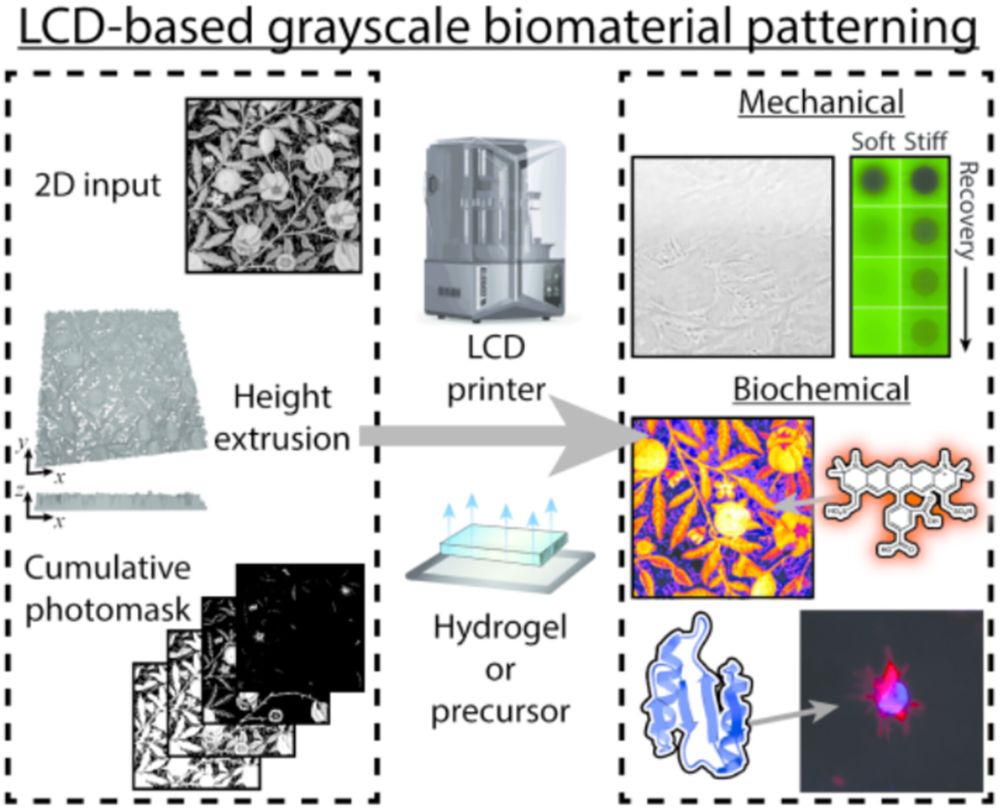
Stimuli-responsive biomaterials hold great promise in controlled therapeutic delivery, tissue engineering, and biosensing applications. Recently, molecular assembly via autonomous compilation has been employed to create topologically specified protein cargos that can be site-specifically tethered to and conditionally released from biomaterials following user-programmable Boolean logic. Prior implementation has been confined to simple fluorescent protein outputs and model protease inputs. In this manuscript, we extend the applicability of this framework by assembling all 7 unique logical operations emanating from a YES/OR/AND 3-input operator set to deliver bioactive proteins spanning diverse categories: growth factors (epidermal growth factor), model enzymes (β-lactamase, NanoLuciferase, and thioredoxin A), therapeutic nanobodies (anti-human epidermal growth factor receptor 2), de novo-engineered cytokines (Neoleukin), and fluorescent proteins (mGreenLantern). In so doing, we demonstrate programmable biomacromolecule release from material anchors in response to precise combinations of three orthogonal protease actuators while maintaining native bioactivity. Through inclusion of a photocleavable protein motif, we further establish that visible light can be employed as an additional input in specifying logic-based protein release. We anticipate these methods will powerfully expand opportunities for targeted therapeutic delivery and beyond.
Check out our new paper entitled "Boolean Logic-based Controlled Release of Bioactive Proteins with Diversified Inputs", live in @angewandtechemie.bsky.social !
Growth factors, enzymes, nanobodies, and cytokines can now be "logically" delivered!
📜: onlinelibrary.wiley.com/doi/10.1002/...
21.11.2025 21:17 —
👍 8
🔁 2
💬 0
📌 1

Programmable proteins use logic to improve targeted drug delivery
Targeted drug delivery is a powerful and promising area of medicine. Therapies that pinpoint precise areas of the body can reduce the medicine dosage and avoid potentially harmful “off target”...
Medicine isn’t just about finding the right therapies — it’s also about getting them to where in the body they’re needed. These new smart proteins can autonomously read bodily cues to zero in on the exact locations of diseases. @coledeforest.bsky.social
21.10.2025 17:23 —
👍 7
🔁 5
💬 1
📌 0

Protein-based biomaterials have risen in popularity in recent years owing to their genetic encodability, sequence specificity, monodispersity, and ability to interface with biological systems in comparison with synthetic polymer-based materials. Though naturally derived and minimally engineered proteins have been at the forefront of these efforts, recent advances in computational protein design offer exciting opportunities for next-generation biomaterial development. In this work, we employ de novo protein design methodologies to generate a suite of self-assembling multimeric proteins, whose step-growth heteropolymerization into bulk hydrogels and condensates can be exogenously triggered through small-molecule addition. Our results highlight how changes in programmed multimer valency and their triggered assembly yield materials with varying structures and viscoelasticity. We anticipate that these approaches will prove useful in rapidly generating large libraries of stimuli-responsive biomaterials that are precisely tailored to specific applications in the biosciences and beyond.
Our latest collaborative paper with David Baker's lab is now live in Cell Biomaterials (@cp-cellbiomat.bsky.social, Cell Press)! Check out "Stimuli-triggered Formation of De Novo-designed Protein Biomaterials"!
🔓 : www.cell.com/cell-biomate...
20.10.2025 22:46 —
👍 28
🔁 4
💬 0
📌 0

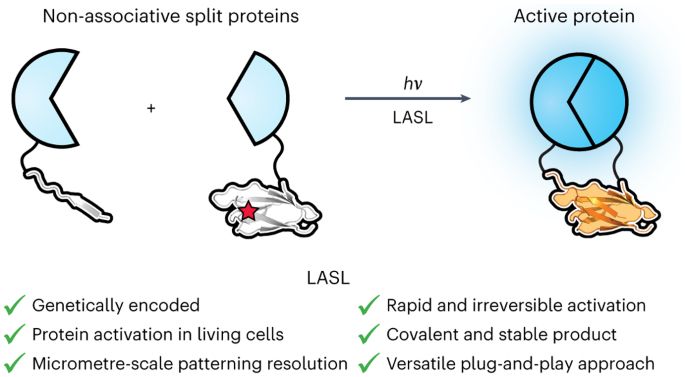
Stimulus-responsive materials have enabled advanced applications in biosensing, tissue engineering and therapeutic delivery. Although controlled molecular topology has been demonstrated as an effective route toward creating materials that respond to prespecified input combinations, prior efforts suffer from a reliance on complicated and low-yielding multistep organic syntheses that dramatically limit their utility. Harnessing the power of recombinant expression, we integrate emerging chemical biology tools to create topologically specified protein cargos that can be site-specifically tethered to and conditionally released from biomaterials following user-programmable Boolean logic. Critically, construct topology is autonomously compiled during expression through spontaneous intramolecular ligations, enabling direct and scalable synthesis of advanced operators. Using this framework, we specify protein release from biomaterials following all 17 possible YES/OR/AND logic outputs from input combinations of three orthogonal protease actuators, multiplexed delivery of three distinct biomacromolecules from hydrogels, five-input-based conditional cargo liberation and logically defined protein localization on or within living mammalian cells.
Our new paper entitled “Boolean Logic-gated Protein Presentation through Autonomously Compiled Molecular Topology” is now live at Nature Chemical Biology (@natchembio.nature.com)! Check it out!
📜: www.nature.com/articles/s41...
@uwnews.uw.edu feature write-up: www.washington.edu/news/2025/10...
13.10.2025 16:22 —
👍 16
🔁 4
💬 0
📌 1
Mike Regnier, Farid Moussavi-Harami, Nate Sniadecki, and their respective labs also played instrumental roles on these exciting efforts!
12.09.2025 19:46 —
👍 0
🔁 0
💬 1
📌 0
This has been a huge collaborative effort overseen by Jennifer Davis and driven by co-first authors Ross Bretherton (co-advised between the DeForest and Davis labs) and Bella Reichardt.
12.09.2025 19:46 —
👍 0
🔁 0
💬 1
📌 0
Congrats, @nrichbourg.bsky.social ! Looking forward to watching your lab grow at FASU-FSU!
20.05.2025 12:02 —
👍 1
🔁 0
💬 0
📌 0
Fantastic! This is such great news. Congrats on the new position, @monicaohnsorg.bsky.social !
20.05.2025 12:00 —
👍 1
🔁 0
💬 1
📌 0

Four UW undergraduates named 2025 Goldwater Scholars
Four University of Washington students have been named Goldwater Scholars by the Goldwater Foundation, advancing the University’s tradition of undergraduate academic excellence and...
For the 2nd year in a row, one of our lab members has been selected for the prestigious Goldwater Scholarship. Huge congrats to Shivani Kottantharayil for her 2025 selection (and to Annabella Li for receiving the award last year)!
UW press release is here: www.washington.edu/uaa/2025/04/...
13.05.2025 19:50 —
👍 5
🔁 0
💬 0
📌 0


Happy Holidays from the DeForest Lab!
What fun to celebrate the group’s 2024 achievements, especially in the baking sector. Our annual dessert bake-off competition was a tasty success!
12.12.2024 20:41 —
👍 12
🔁 0
💬 0
📌 0
The Anseth lab continues to pioneer many of the best and most versatile photoresponsive biomaterials known to our community. Congrats to @monicaohnsorg.bsky.social and Ella Hushka for summarizing the group's last 15 years of exciting efforts in their new paper!
12.12.2024 17:45 —
👍 11
🔁 2
💬 0
📌 0
Fun story from @matthewfainor.bsky.social on how next-gen recombinant proteins are being expressed in fruit flies, and what implications that could have for our lab-grown hydrogel biomaterials!
04.12.2024 16:03 —
👍 6
🔁 1
💬 0
📌 0

Professors often tell graduate students that “Your PhD is a #marathon, not a sprint”.
Having now run the 2024 Seattle Marathon (many years after finishing a PhD), I now actually feel qualified to make such a statement.
@uwmedicine.bsky.social
02.12.2024 21:16 —
👍 25
🔁 0
💬 0
📌 0
Would love to be included here! Thanks for organizing!
21.11.2024 04:59 —
👍 1
🔁 0
💬 1
📌 0

HUB
Introducing the Humanity Unlocking Biomaterials (HUB) national coordinating center. Opportunity to become involved with an incredible community, $100k research grants, core resources, and much more. Come join us! See more at our new website, here: www.humanityunlockingbiomaterials.org
13.11.2024 17:39 —
👍 32
🔁 14
💬 1
📌 2
Thank you, on all accounts!
12.11.2024 15:08 —
👍 1
🔁 0
💬 0
📌 0
Great idea! We work in this space!
12.11.2024 04:45 —
👍 1
🔁 0
💬 1
📌 0
Great idea! Thanks for helping coordinate this transition!
10.11.2024 13:35 —
👍 2
🔁 0
💬 1
📌 0
Great idea! Glad to see the biomaterials community starting to come together here on Bluesky.
10.11.2024 13:34 —
👍 4
🔁 0
💬 0
📌 0